Omega-3 supplements accelerated cognitive decline in older adults, study finds
A 2026 analysis of over 800 older adults found omega-3 supplement users declined faster on three cognitive measures than matched non-users. Reduced brain glucose metabolism, not amyloid buildup, appeared to drive the effect.
Omega-3 fatty acid supplements, among the most widely consumed dietary supplements on the planet, were associated with faster cognitive decline in a longitudinal analysis of more than 800 older adults published in 2026. That finding lands against the grain of decades of observational work suggesting fish oil protects the aging brain — and it held after the researchers matched users and non‑users on a broad set of demographic, genetic, and health variables.
Conducted by a team at the Army Medical University in China and published in The Journal of Prevention of Alzheimer’s Disease, the study drew on data from the Alzheimer’s Disease Neuroimaging Initiative (ADNI), a long‑running, multi‑site cohort that tracks older adults with repeated cognitive assessments, brain imaging, and biomarker collection. From this pool, the researchers identified 273 individuals who reported long‑term omega‑3 supplement use and matched them to 546 non‑users with similar baseline cognition, age, education, and APOE‑ε4 carrier status.
The result was not subtle.
Across a median follow‑up of roughly five years, the supplement group deteriorated faster on all three cognitive instruments the study tracked: the Mini‑Mental State Examination (MMSE), the Alzheimer’s Disease Assessment Scale–Cognitive Subscale (ADAS‑Cog), and the Clinical Dementia Rating (CDR). All differences reached statistical significance and, the authors noted, were clinically meaningful. On the MMSE alone, the supplement group lost roughly 0.8 more points per year than matched controls — a gap that compounds across half a decade.
What made this finding different
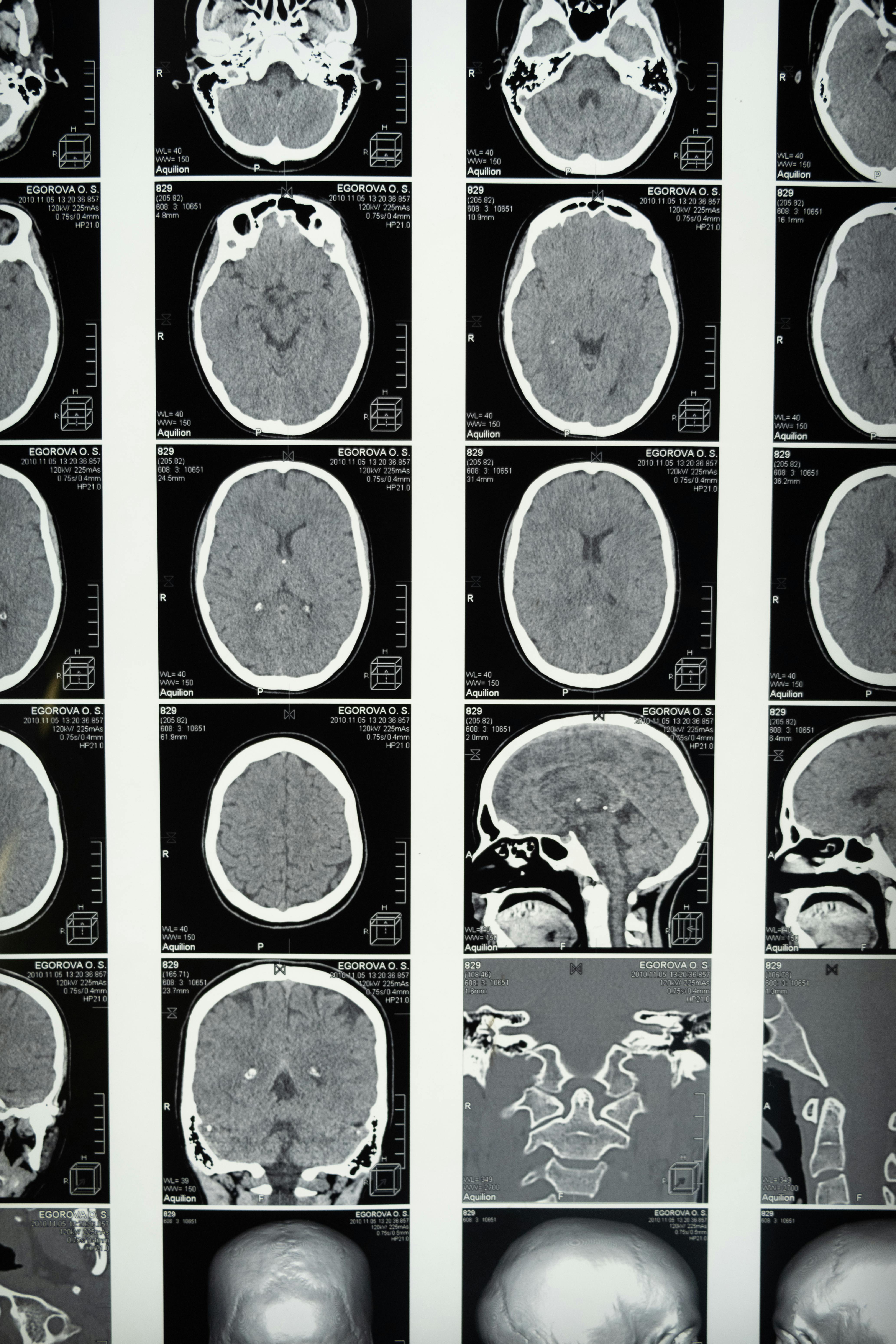
Most omega‑3 brain studies have been either short‑term randomized trials, where participants take fish oil for six to 24 months while researchers track cognitive endpoints, or population cohorts that link dietary intake questionnaires to dementia incidence years later. But the ADNI‑based approach is different. It layers supplement‑use history onto a dataset that already contains serial cognitive testing, amyloid and tau PET imaging, structural MRI, and fluorodeoxyglucose (FDG‑PET) scans — a measure of how much glucose the brain is burning in different regions.
That imaging depth is what allowed the researchers to ask a question earlier studies could not: if omega‑3 users are declining faster, what is actually happening inside their brains?
And yet the pathology scans told a story nobody expected. Amyloid plaque burden was no different between the groups. Neither was tau deposition or overall brain atrophy. Structurally, the two groups looked indistinguishable on the markers that typically track Alzheimer’s progression. So the FDG‑PET data became the paper’s most important thread.
The glucose hypothesis
Supplement users showed reduced cerebral glucose metabolism in regions known to be vulnerable in early Alzheimer’s disease: the posterior cingulate, precuneus, and lateral temporal and parietal cortices. These are the same regions that dim on FDG‑PET scans years before a person develops clinical symptoms. In a mediation analysis, reduced glucose uptake in these areas accounted for roughly 30 to 40 percent of the accelerated cognitive decline seen in the omega‑3 group.
Crucially, the authors are careful about what this means. They do not claim omega‑3 causes brain damage. Their hypothesis, laid out in the discussion, is more specific: high intake of polyunsaturated fatty acids may alter synaptic membrane composition in ways that reduce neuronal fuel efficiency. DHA, in particular, is highly incorporated into synaptic membranes. In excess, the authors suggest, it might shift membrane fluidity past a functional optimum, affecting the trafficking of glucose transporters or the efficiency of mitochondrial respiration at the synapse.
Granted, it is a mechanistic argument, not a proven pathway. But it is biologically plausible, and it explains the otherwise puzzling disconnect between normal amyloid scans and faster clinical decline. Prior work cited in the paper shows that DHA‑enriched neuronal membranes are more susceptible to lipid peroxidation — an oxidative process that can damage membrane‑bound proteins, including glucose transporters. If that damage accumulates faster than the cell can repair it, synaptic energy metabolism suffers. What looks like a protective lipid on a supplement label may, in some aging brains, become a substrate for oxidative stress.
This is not the first signal that omega‑3 supplementation might cut both ways. A 2025 systematic review found that low doses of fish oil — in the range of 400 to 800 mg of combined EPA and DHA per day — were associated with small cognitive benefits in some trials, but daily intakes above roughly 1,500 mg reversed any advantage. Nor are the cardiovascular benefits immune from this pattern: protective at moderate intake from food, neutral or even pro‑arrhythmic at high supplemental doses in certain populations.
What the study cannot tell us
Because the analysis is observational, the two groups may have differed in ways the matching did not capture. Supplement users in the ADNI cohort were, on average, more educated and more likely to be white and female than non‑users. Statistical controls for these variables were applied, and the results persisted across multiple sensitivity analyses — but residual confounding is always possible when a study design does not randomize.
Nor can the dataset distinguish between types of omega‑3 supplements — fish oil versus algal DHA versus krill oil — or between doses. Participants were classified as users or non‑users based on self‑report, and detailed dosing histories were not available. A person taking 300 mg of DHA from algae twice a week is folded into the same category as someone taking 2,000 mg of fish oil daily. That matters because the dose‑response relationship, if one exists, is almost certainly not linear.
Generalizing these findings to younger adults or to people taking omega‑3s for cardiovascular reasons is also premature. The study population was already at elevated risk for cognitive decline given their enrollment in ADNI. Still, the results demand attention because they land in a field where supplementation is widespread and largely unquestioned. As we covered in our broader evidence review, global omega‑3 supplement sales exceed $5 billion annually, and surveys consistently find that “brain health” is among the top reasons consumers give for taking fish oil.
Where this leaves the evidence
None of this overturns the omega‑3 literature. It complicates it. Epidemiological evidence — people who eat fish regularly tend to have lower dementia risk in cohort studies — has always been the strongest card for brain health, but those associations may reflect the broader dietary pattern of fish‑eaters rather than a direct effect of the fatty acids themselves. Randomized trials of fish oil supplementation for cognitive outcomes have been largely null, a fact the supplement industry rarely highlights.
What the new study adds is a specific, testable hypothesis: that omega‑3 supplementation at common doses may, in some older adults, reduce synaptic glucose metabolism. Confirmed in a dedicated trial with pre‑ and post‑supplementation FDG‑PET imaging, that finding would force a re‑evaluation of the supplement’s risk‑benefit calculus for brain health.
Until then, the practical implication is narrower than the headlines suggest. This was not a trial. It did not randomize anyone to fish oil. And it does not mean that eating salmon is harmful — food‑sourced fatty acids, by contrast, consistently show protective associations, arriving in a matrix of protein, selenium, and vitamin D that supplementation strips away.
What it does mean is that the assumption that omega‑3 capsules are harmless to the aging brain has lost its footing. For older adults taking high‑dose fish oil specifically for cognitive protection, the evidence base now runs in both directions, and the more granular the imaging, the harder the protective story is to sustain.
References
- Research Team at Army Medical University. Association of long‑term omega‑3 supplement use with accelerated cognitive decline in older adults: a longitudinal analysis of the ADNI cohort. The Journal of Prevention of Alzheimer’s Disease. 2026. https://www.sciencedirect.com/science/article/pii/S2274580726000932
- Sydenham E, Dangour AD, Lim WS. Omega‑3 fatty acids for the prevention of dementia. Cochrane Database of Systematic Reviews. 2023. https://doi.org/10.1002/14651858.CD005379.pub4
Cognitive science writer covering nootropics, focus protocols, and the evidence behind brain supplements. Reports from Stockholm.
The Vitalspell brief
Evidence-based supplement science — weekly in your inbox.
Subscribe

